I wrote a check for $50,000 into this Company.
That’s because I believe that this $50,000 could soon be worth many times that.
In fact, in just a bit, I’ll show you why a 15,700% potential return is possible from here.
And it all starts with a very special situation that could soon propel the shares of this Company.
It’s one that involves one of the smartest scientists in North America and her breakthrough in a multi-billion dollar, life-changing industry.
It’s why she was named one of the most powerful women last year.
Yet, very few investors know about this Company – but I believe very strongly that many soon will.
This isn’t just a random breakthrough.
This Company has over 20 years of research, more than $25 million spent on R&D, and patents in both N. America and Europe for its breakthrough discovery.
This technology is a first-of-its-kind and has the potential to change an industry worth billions upon billions of dollars.
And it’s about to transition from the lab to the real world.
That could mean serious profits for a company with a market cap of barely over CDN$30 million – incredible considering the amount of research and money already poured into this technology.
It also means shareholders could soon be rewarded as well.
It’s no wonder why big names are already jumping at the chance to get involved.
In fact, one big pharmaceutical company just invested $1 million into this tiny junior and has already signed an exclusive license and distribution agreement to lock down this tech in certain parts of the world.
But big profits are just a small benefit to what this Company could soon achieve.
This Company has the potential to change lives.
Serious as Cancer
Chances are you or someone you know is going to get cancer.
I don’t mean to sound harsh, but 40% of us will be diagnosed with cancer at some point during our life.
In the U.S. alone, The National Cancer Institute estimates that about 595,690 Americans will die of cancer this year. That’s more than one every minute.
But this should come as no surprise.
I am sure many of you have already gone through a very difficult time in dealing with cancer – whether it was your own or someone you love.
I know I did.
A Most Difficult Decision
That brings me to a very difficult subject.
Anyone who has had to deal with cancer knows that one of the most difficult decisions we can make is how we deal with it.
That’s because after a cancer diagnosis, patients and their families have to make a number of decisions about treatment.
How do we know what treatment is right?
Do we need radiation?
Do we need surgery?
Do we need chemotherapy?
How do we know if one treatment is better than another?
How do we know if a treatment is actually working?
There isn’t a simple answer for any of these questions because there is no “one size fits all” treatment for any cancer.
Unfortunately, that means many diagnosed with cancer are often mistreated, or worse, over-treated.
According to the Prostate Cancer Foundation:
“…current estimates indicate that 30% more men are aggressively treated for prostate cancer than is necessary to save a life from the disease.”
This not only costs healthcare payers billions of dollars every year, but it often inflicts unnecessary treatment and complications on patients.
For example, chemotherapy and radiation are often used to fight cancer – both of which can take a serious toll on a patient.
Here is just one story of a women named Hazel…
Via Cancer Tutor:
“Two years ago, Hazel was diagnosed with breast cancer. She described her chemotherapy as the worst experience of her life.
“This highly toxic fluid was being injected into my veins. The nurse administering it was wearing protective gloves because it would burn her skin if just a tiny drip came into contact with it. I couldn’t help asking myself, ‘If such precautions are needed to be taken on the outside, what is it doing to me on the inside?’
“From 7 p.m. that evening, I vomited solidly for two and a half days. During my treatment, I lost my hair by the handful, I lost my appetite, my skin color, my zest for life. I was death on legs.”
She is not alone in her experience. Nearly everyone who undergoes such harsh treatments has similar stories.
Now imagine if it turned out that after the chemotherapy, her cancer didn’t die.
Imagine if we later found out that another treatment would have been the better option?
That’s just the tip of the iceberg:
“…Some 67 percent of people who die during cancer treatment do so through opportunistic infections arising as a direct result of the immune system failing because of the aggressive and toxic nature of the drugs.”
In other words, some cancer treatments can even lead to death.
Yet, despite all of this, there hasn’t been an accurate way to tell what treatment is best for those diagnosed with cancer. Nor have there been ways to determine how a patient is reacting to treatment in real time.
That’s why one of the biggest challenges for cancer physicians is recommending the right treatment.
No physician wants to be responsible for mistreating a patient.
But what if it didn’t have to be this way?
What if there was a technology that could more accurately inform you about treatment?
What if there was a technology that could not only determine how your disease is progressing, but also how you’re responding to treatment in real time?
Why go through the perils of radiation, chemotherapy, or surgery if it might not work?
Anyone who has ever had to deal with cancer knows just how significant this could be.
After 20 plus years of research, more than $20 million in R&D, more than 100 peer-reviewed papers, and 16 clinical studies, one of 2015’s most powerful Canadian woman is about to bring such a technology to the world.
CONTENT LOCKED
Enter your email to get instant access (it's free!)
to this special content post:
*By entering your email, you are agreeing to our privacy policy and terms of use. You will also receive a free weekly subscription to the Equedia Letter, one of Canada's largest private investment newsletters. Don't worry, it's free and you can cancel at anytime.
Her name is Dr. Sabine Mai and her breakthrough research is finally moving from the bench to the bedside to enable better treatment and monitoring options for patients.
That’s why she co-founded the company I am about to introduce.
And that’s why the value of this Company could soon soar.
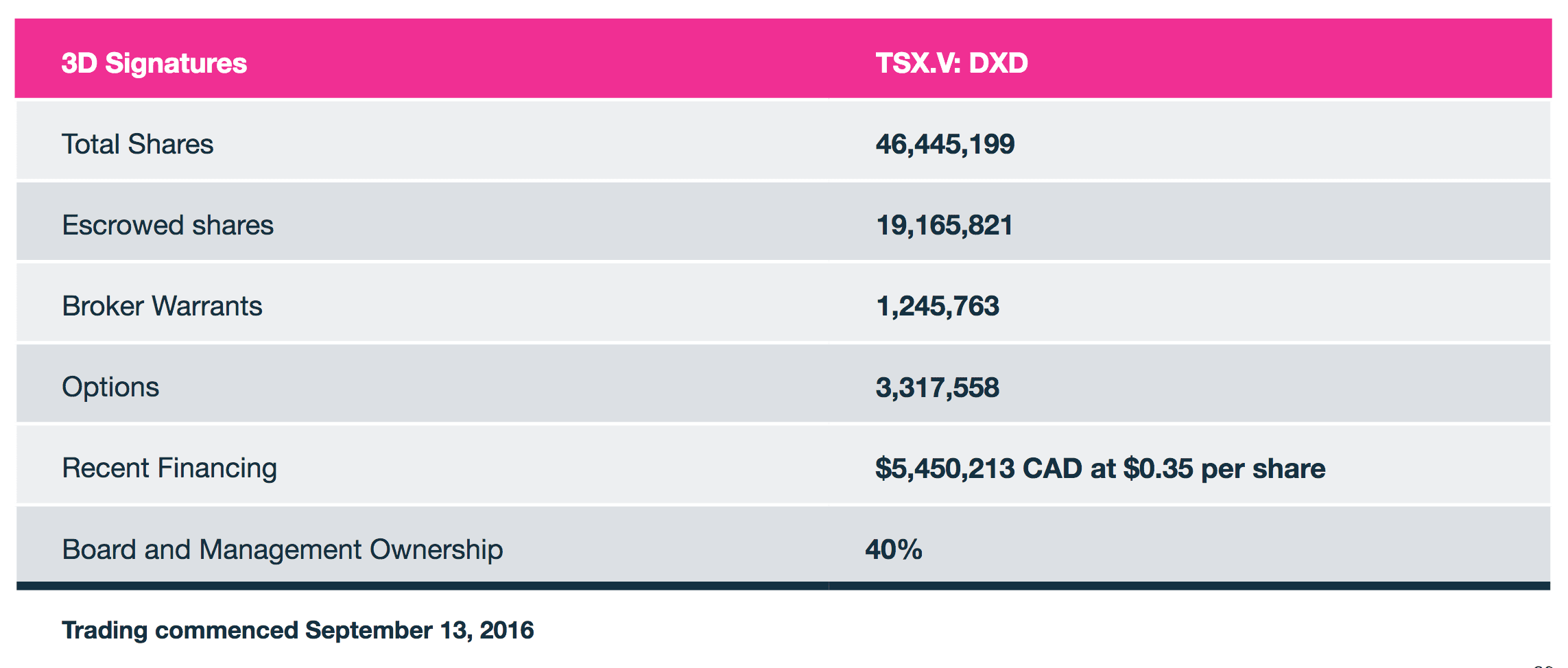
3D Signatures Inc.
Canadian Trading Symbol: TSX-V: DXD
US Trading Symbol: OTCQB: TDSGF
German Trading Symbol: FSE: 3D0
An Entirely New Class of Biomarkers
3D Signatures Inc. (3DS) has discovered an entirely new class of biomarkers for diagnosis, assessment, and monitoring of major diseases, with a current focus on Cancer and Alzheimer’s Disease.
The discovery of a new biomarker alone represents a major breakthrough.
A biomarker is “a measurable substance in an organism whose presence is indicative of some phenomenon such as disease, infection, or environmental exposure.”
According to a new report by Grand View Research Inc., the global biomarkers market is expected to reach over US$78.2 billion by 2024.
And according to market research firm Kalorama, the cancer biomarker is expected to reach over US$7.4 billion by 2020.
But 3DS has gone far beyond just identifying a new biomarker.
Using this new biomarker, 3DS has developed precision clinical tests that are powered by a proprietary software platform that can measure:
- the stage of disease
- the rate of progression of a disease
- drug efficacy
- and drug toxicity
That means instead of “guessing” what treatment to use based on “guessing” the stage of disease and “guessing” how it’s progressing, we no longer have to “guess.”
It potentially means once we select a treatment, we can know right away if it’s working, see if there are any adverse side effects, and know if the treatment is doing more harm than good – all in near real-time.
In fact, the technology is designed to predict the course of a disease and customize treatment for each individual patient.
This has never been done before.
All of this is backed by more than 20 years of research and has already been validated in numerous papers, supported by 16 successful clinical studies on over 1,500 patients on 13 different cancers (including prostate, breast, lung and multiple myeloma), and Alzheimer’s disease.
This type of personalized treatment is truly groundbreaking and could soon be adopted by clinics everywhere.
For a company worth just over CDN$30 million, the upside could be tremendous.
I’ll get back to this in a bit.
But before I do, it’s important to understand how it works.
How 3DS Technology Works
Every human has DNA that is packaged into chromosomes.
At the tip of each chromosome are protective regions of DNA called telomeres.
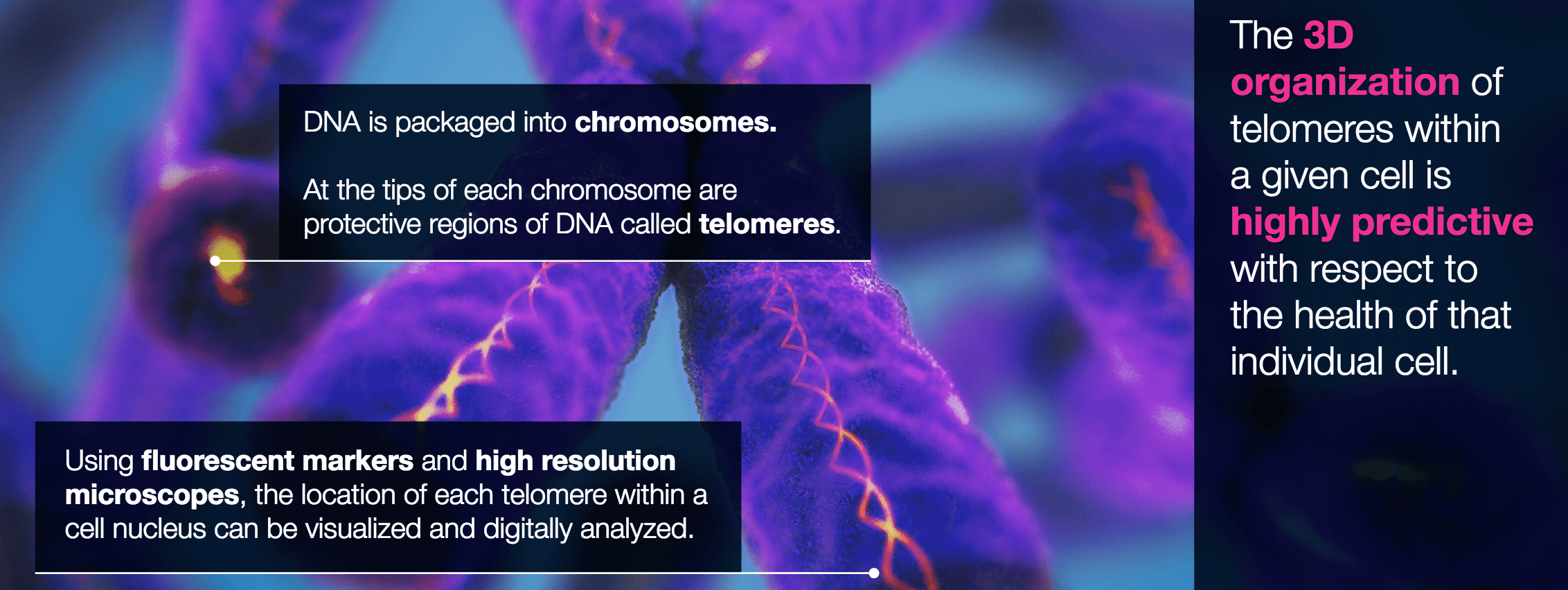 By using fluorescent markers and high-resolution microscopes, we can see the location of each telomere within a cell nucleus and digitally analyze it.
By using fluorescent markers and high-resolution microscopes, we can see the location of each telomere within a cell nucleus and digitally analyze it.
3DS patented software platform then analyzes an individual’s three-dimensional chromosomal structure or “Signature” and specifically analyzes the patient’s telomeres in a three-dimensional manner.
This three-dimensional organization of telomeres within a cell nucleus is highly predictive with respect to the health of that individual cell.
In other words, we can tell a lot just by looking at the health and shape of these telomeres.
So how does that help us?
- Screening – Who has a particular disease?
- Diagnosis – What kind of disease/cancer?
- Prognosis – How stable or aggressive is the disease/cancer?
- Predicting – How will the patient respond to a particular treatment?
- Monitoring – Stability/progress of patient over course of treatment
- Drug Development – Identification/development of better drugs
Real-Life Scenario
Depending on the disease, a clinic takes a tissue, blood, or a cheek swab sample.
This sample is then preserved and labeled.
Pictures of these samples are then taken from multiple 3D perspectives, which are then exported to a centralized 3D Signatures analytics centre.
Once at the centre, the images are analyzed and then given a scoring model based on the parameters generated by 3DS technology.
A personalized report is finally generated and sent back to the clinic with clear information for the patient.
It’s a super simple, non-invasive process.
With 3DS, we can now accurately provide a host of predictive medical information in real-time, using nothing more than blood, cheek swab or tissue samples.
The impact and benefits of this technology are truly significant.
Helping medical professionals deliver the best care protocols for each patient will not only benefit patients immensely, but it could save health care providers billions of dollars by reducing ineffective, improper, and unnecessary treatments.
Imagine what it could save for insurance companies.
The potential market for this alone is staggering.
But just how big is the market?
Creating Beachhead
As of right now, 3DS trials include 14 diseases and 16 clinical trials on over 1500 patients.
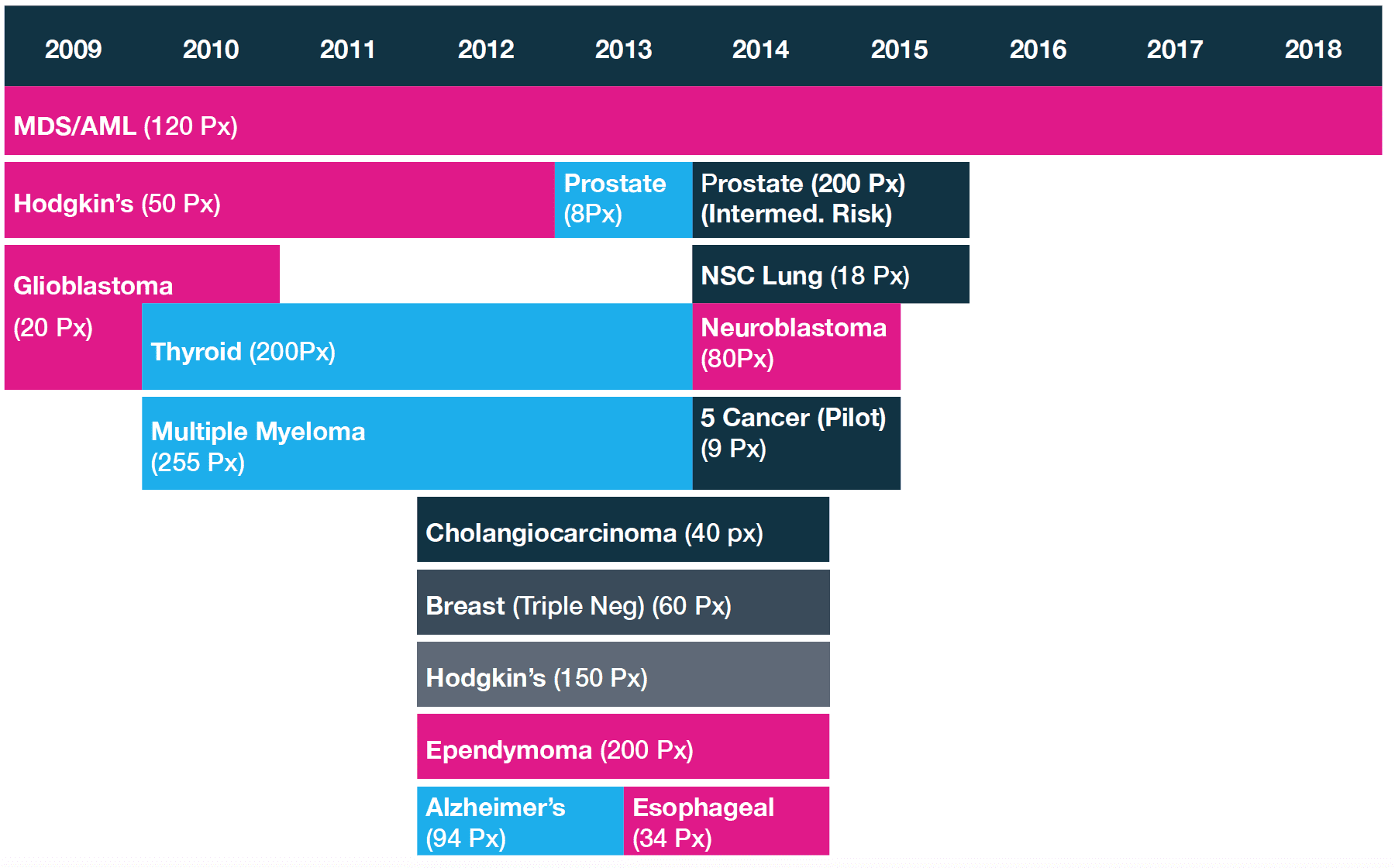
Here is the pipeline and stage of each niche for 3DS technology:

As you can see, 3DS works on a wide spectrum of diseases, with many of them already moving into the development and validation stage. In fact, there is potential to work on even more diseases and cancers.
But for now, let’s focus on the two that are closest to market.
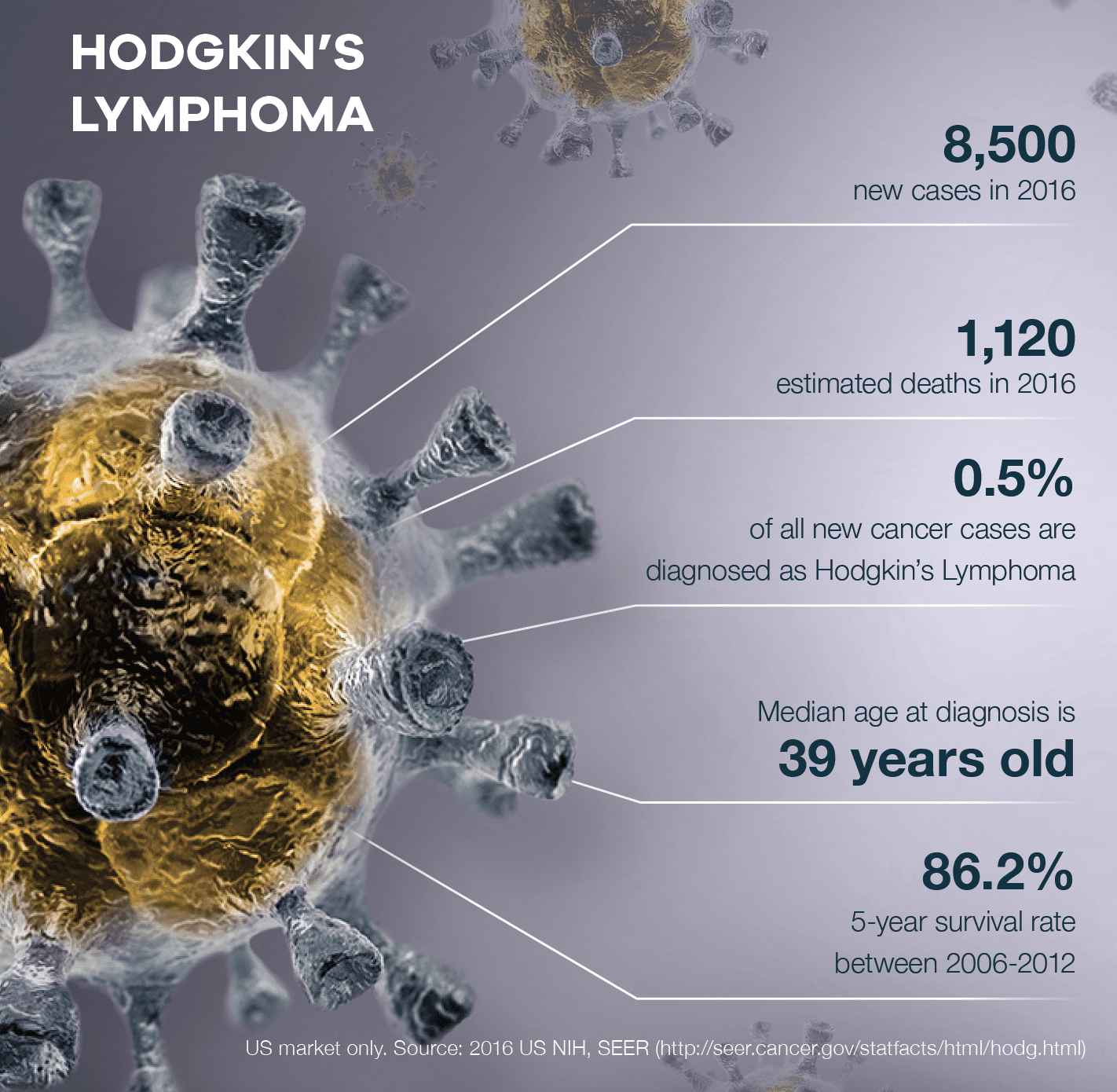
Hodgkin’s Lymphoma
For almost all patients with Hodgkin disease, cure is the main goal.
The 2 main ways of treating Hodgkin disease are chemotherapy and radiation therapy. Depending on the situation, one or both of these treatments might be used.
However, these treatments can have side effects that often don’t show up for many years. Because of this, doctors try to choose a treatment plan with the lowest risk of possible side effects.
But since physicians can’t predict whether a patient will respond to standard chemotherapy and enter long-term remission or not respond and relapse, treatment choices become even more difficult to make.
It also means that patients have no way of knowing if the treatments are working until long after it’s over.
By that time, the treatments could have done more harm than good.
There simply is no test capable of providing clinicians with the type of information for proper treatment in real-time.
Except for 3D Signature’s platform.
3DS is designed to determine whether an individual will respond to standard chemotherapy almost immediately.
Once a treatment is selected, it can also determine if the treatment is working in real time.
Those who are shown to not respond to chemotherapy can then receive optimized alternative treatments resulting in:
- New treatment options
- Reduced secondary complications
- Significant cost savings
This is truly groundbreaking when it comes to treating those diagnosed with Hodgkin’s Lymphoma.
But as significant as this is, Hodgkin’s Lymphoma is a small market when compared to other cancers.
Such as prostate cancer…
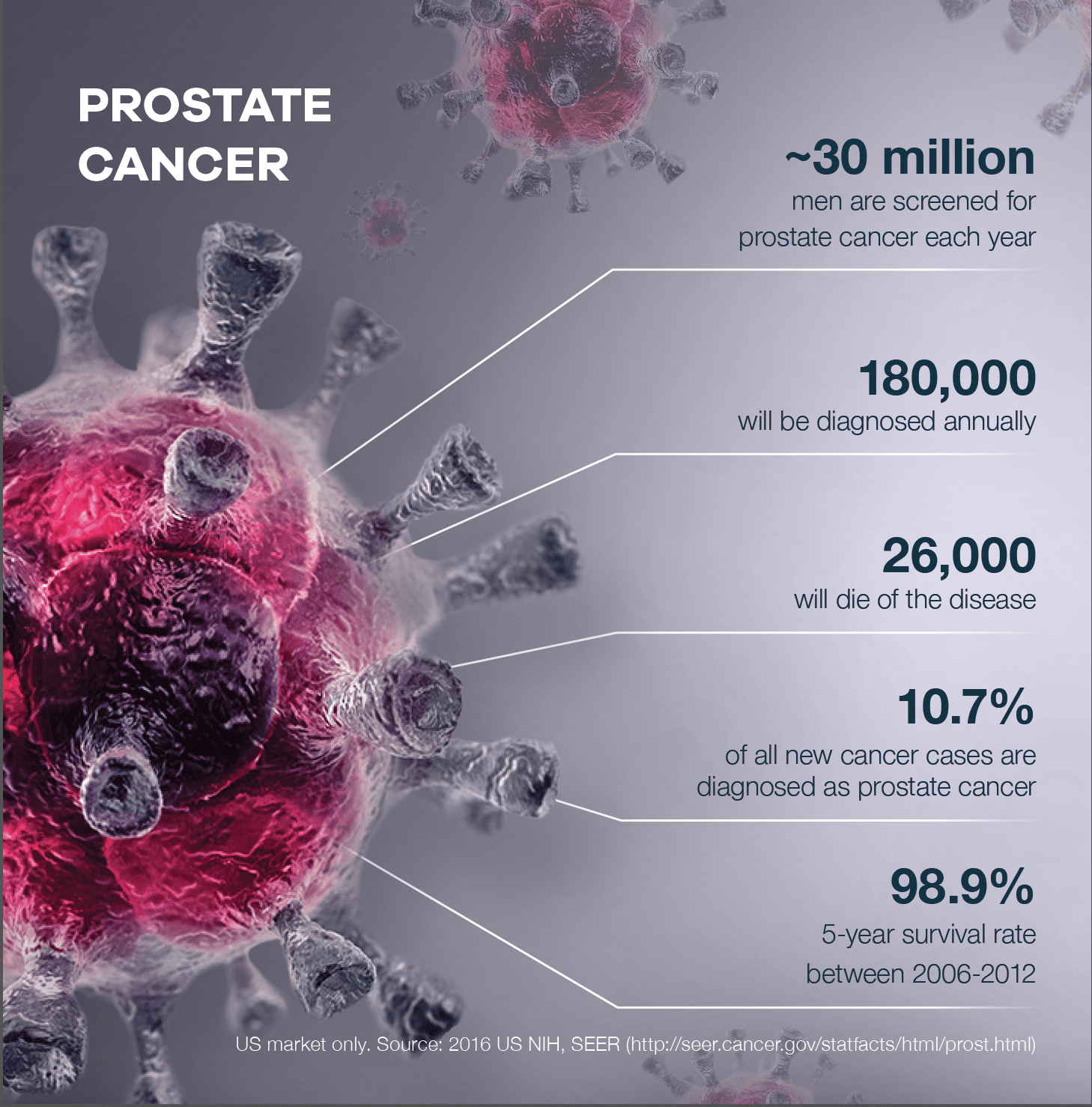 Prostate Cancer
Prostate Cancer
One in seven men will be diagnosed with prostate cancer during his lifetime.
And one in 39 men will die from it.
In fact, there are over three million existing and new cases of prostate cancer in the United States and Canada alone.
As I mentioned earlier, many diagnosed with prostate cancer are often mistreated because of the dangers that might arise if left untreated.
If we could avoid over-treating the 80% of men with low-grade disease who will never die of prostate cancer, we would save billions of dollars every year.
More importantly, thousands of men may not have to suffer some of the dangerous treatments involved in treating prostate cancer.
Right now, there is no test to accurately determine which intermediate-risk patients will progress to an advanced cancer and require immediate aggressive treatment.
But 3DS technology is designed to stratify patients into several risk categories and predict the most effective treatment plan.
And best of all, the test is a non-invasive risk assessment and monitoring platform based on a blood sample, and not a prostate biopsy.
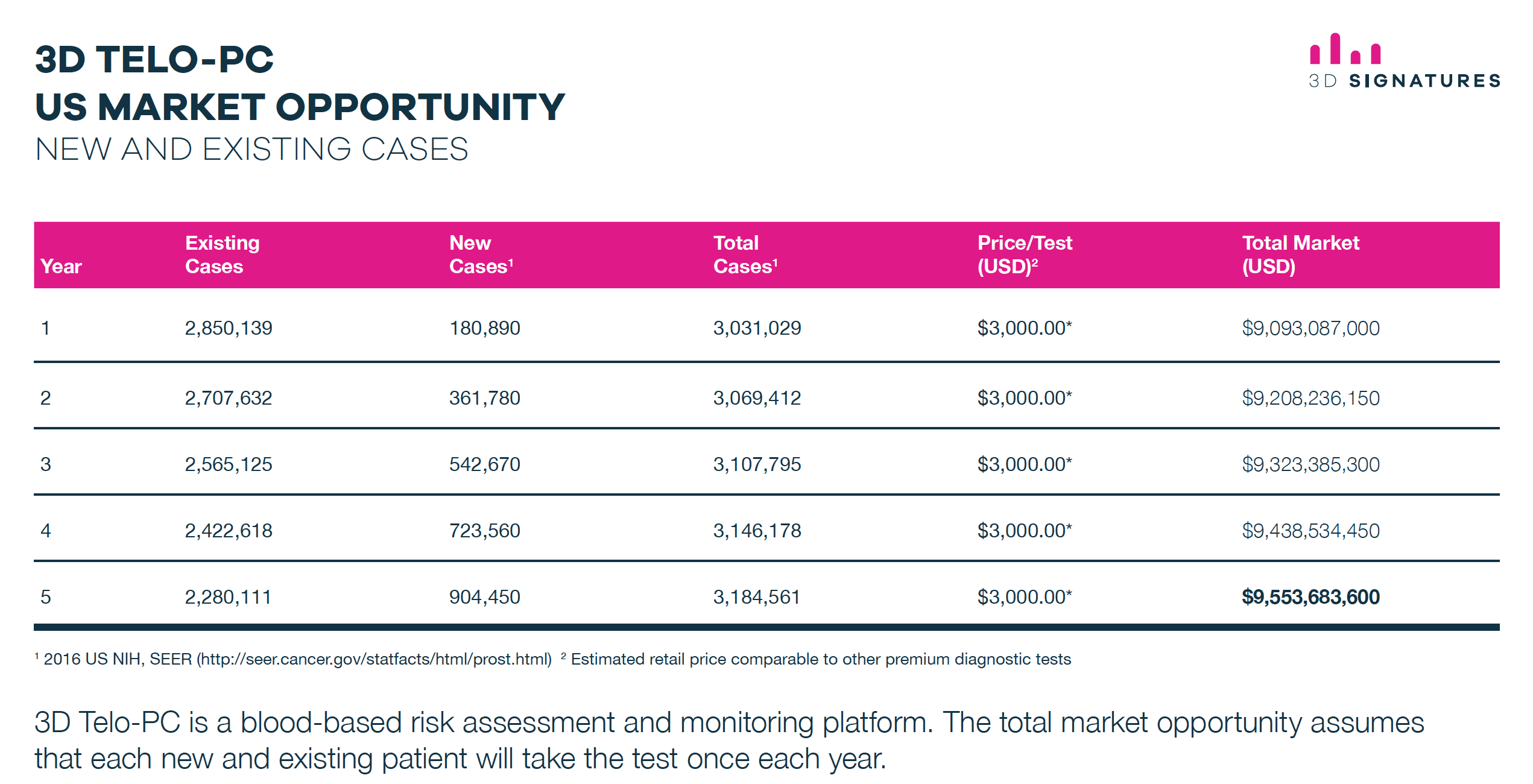
US$9 Billion-Dollar Prostate Cancer Market
Based on an estimated retail price of US$3,000 per 3DS Telo-PC test, the total market revenue could be over US$9 billion per year!
Let’s put that into perspective.
If 3DS captures just 10% of the prostate test market, that’s nearly US$1 billion in revenue.
3DS is worth barely over CDN$30 million as I write this.
The average price to sales ratio of those in the healthcare information and technology space is 3.54.
Using back-of-the-napkin math, based on the 3.54 ratio, the current US-CDN exchange rate of 1.34, current 3DS share structure, and assuming 3DS captures 10% of the prostate test market, it means 3DS could be worth over CDN$96 per share!
That’s a potential return of more than 15,700%!
 Of course, take the back-of-the-napkin calculation with a grain of salt.
Of course, take the back-of-the-napkin calculation with a grain of salt.
It will take time for 3DS Telo-PC to fully come to market and there are other risks along the way. The Company will likely dilute further as they raise more money to progress, and going to market involves time and risks.
I am also not saying that once 3DS technology comes to market in full for prostate cancer that everyone will take the test, nor will it be fully adopted by every clinic – that would be very optimistic thinking.
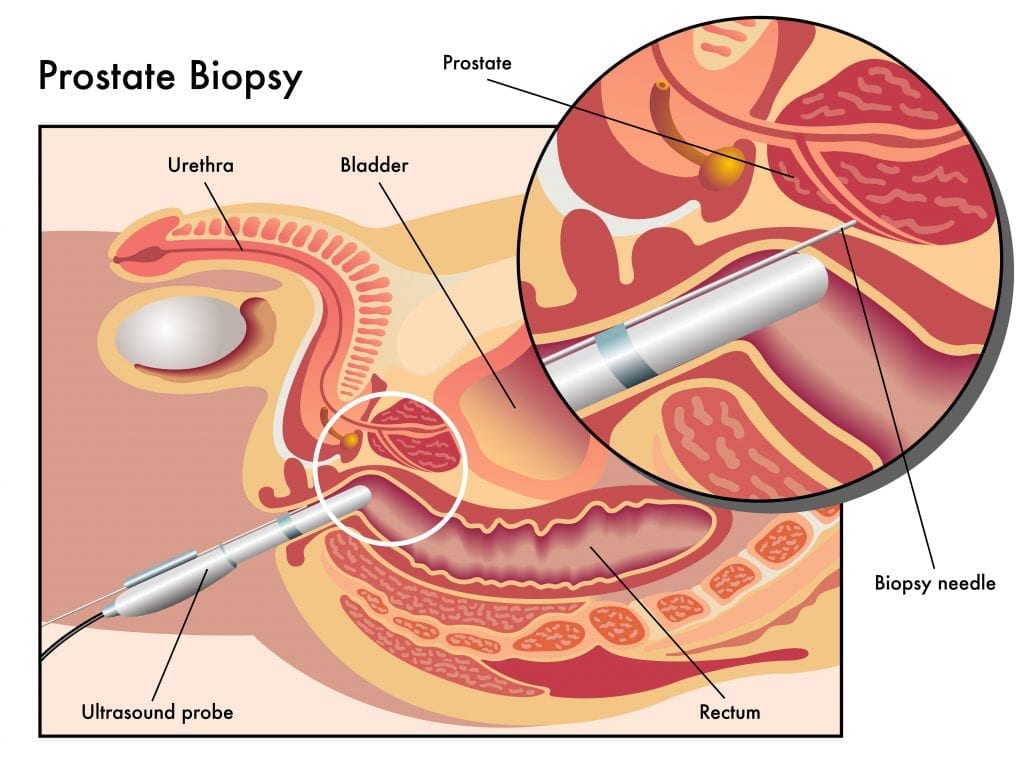 But if you’re a man reading this, would you rather take a non-invasion blood sample or a very invasive prostate biopsy?
But if you’re a man reading this, would you rather take a non-invasion blood sample or a very invasive prostate biopsy?
I wouldn’t think twice about which one I prefer.
And while there are other tests one can do for prostate cancer, 3DS is a dynamic biomarker that can be used throughout the course of the disease and treatment(s).
All of the other tests are static, meaning each test is only relevant for a particular piece of information/stage.
But that’s not all.
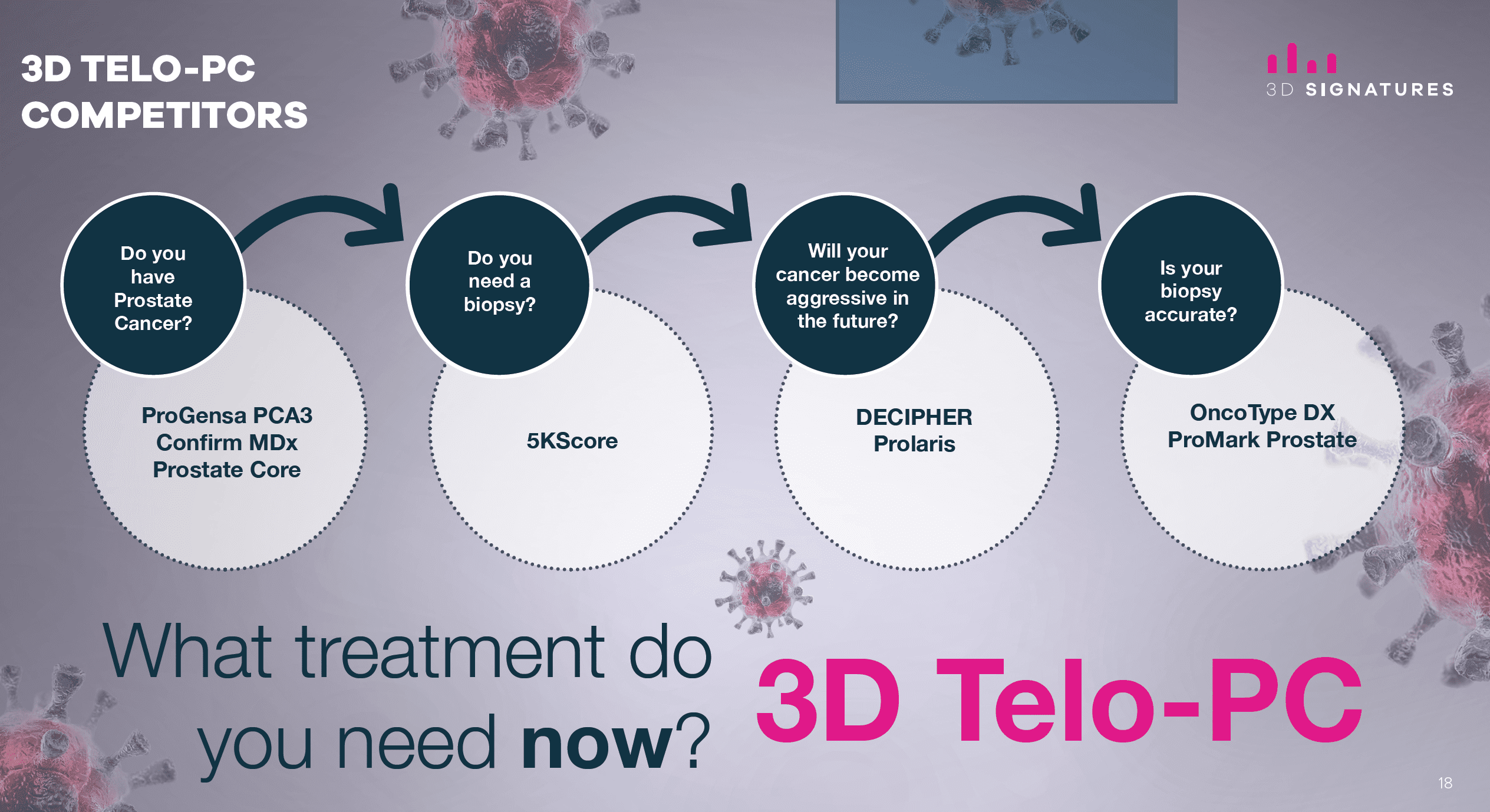 All of the other tests are statistically based, meaning they will provide a probability of progressing within a period of time.
All of the other tests are statistically based, meaning they will provide a probability of progressing within a period of time.
In other words, the other tests are practically a guessing game.
3DS, on the other hand, is a real-time assessment of what stage you’re at and what treatment you need the day you take the test.
It’s a one test fits all.
Lastly, all of the other tests are focused on “the cancer” versus the capacity of dealing with tumour heterogeneity.
Essentially, if you have prostate cancer, you have more than one type of cancer within that tumour.
3DS looks at many circulating tumour cells and can identify and assess multiple tumour subpopulations with the same test.
The implications are significant with respect to understanding the disease and appropriate treatment.
In short, there simply is nothing like 3DS.
That’s precisely why just last week, 3D Signatures Inc. announced that it was one of the few invited to participate in the first randomized, multicenter study focused on biopsy naive patients with clinical suspicion of prostate cancer, known as PRECISE.
3DS Included in One of the Most Significant Clinical Studies in North America for Prostate Cancer
Even more important is what Principal Investigator, Dr. Laurence Klotz, the Chairman of the Canadian Urology Research Consortium and a renowned uro-oncologist and prostate cancer thought leader had to say about 3DS:
“We have an opportunity to test a new blood-based biomarker to accurately stratify patients into risk groups. If successful, this could represent a first-in-class blood test, which would specifically identify clinically significant prostate cancer. Such a tool does not currently exist for prostate cancer patients.”
SUCH A TOOL DOES NOT CURRENTLY EXIST FOR PROSTATE CANCER PATIENTS.
It’s no wonder why 3DS involvement in this study is being funded by the Ontario Institute for Cancer Research and the Movember Foundation Canada:
“Awarded by way of a rigorous peer review process run by Prostate Cancer Canada, this multimillion dollar project is funded by the Ontario Institute for Cancer Research (OICR) and the Movember Foundation Canada. To support its participation in PRECISE and the development of related clinical tests, 3DS’ projected budget is approximately $2.4M over a two-year period which represents at least 50% savings over an independently run study.”
First to Dollar, Then to Regulator
Now here’s the kicker: unlike a drug treatment, 3DS doesn’t have to go through all of the hurdles before it can make money.
It can begin to have clinics using the technology long before full validation of each individual disease – and that means it can begin making money almost immediately.
The best part is that 3DS is just at the beginning stages of roll out to the medical community, which means this is ground-floor investing for a platform that could completely change the way diseases are diagnosed and treated.
But that’s just the tip of the iceberg.
Aside from just clinical tests for patients, which could lead to saving billions of dollars and thousands of lives, there could be something even bigger.
And I am not talking about the billions of dollars in potential revenue for the tests.
In fact, it’s so big and unique that I am not sure how to value it just yet. But I am betting Big Pharma might have an idea.
Let me explain.
Drug Development
Drug development is one of the most costly, time-consuming, and riskiest businesses in the world.
According to a report published by the Tufts Center for the Study of Drug Development (CSDD) back in 2014, the cost of developing a prescription drug that gains market approval is a whopping US$2.6 billion.

Add in post-approval development – studies to test new indications, formulations, and dosage strengths – and costs spike up to nearly US$3 billion.
And, as you can see, the costs have been exponentially rising, year after year.
More importantly, the costs of clinical trials have not only surpassed the costs of preclinical trials but is becoming increasingly more expensive.
That’s because, on average, it takes over a decade to bring a new drug to market.
According to Medicine Net:
“In the United States, it takes an average of 12 years for an experimental drug to travel from the laboratory to your medicine cabinet. That is if it makes it.
Only 5 in 5,000 drugs that enter preclinical testing progress to human testing.
One of these 5 drugs that are tested in people is approved.
The chance for a new drug to actually make it to market is thus only 1 in 5,000.”
So not only is it expensive, it’s extremely time-consuming and extremely risky.
But what if we could shorten this time during the most expensive part of the drug approval, the clinical trial?
You see, a lot of the time spent on the drug approval process is during clinical trials – which is one of the reasons it costs a lot of money.
Clinical trials have many phases, starting with a small group of patients and then more and more as we move through the phases.
During each phase, volunteer patients are monitored to see how they react to the drug.
In other words, wait and see.
But what if drug companies didn’t have to wait and see?
What if they could know in real-time if the drug was working?
What if they could know in real-time if the drug was toxic?
What if they could know in real-time how certain drugs reacted with different individuals?
That’s precisely what 3DS could soon do for Big Pharma.
Based on published work by Dr. Mai and her collaborators regarding real-time ex-vivo feedback on several new therapeutic candidates and inbound interest from a number of major pharmaceutical companies, 3D Signatures is already in discussions around partnership opportunities with some very big names.
That’s because 3DS technology has the potential to identify lead therapeutic candidates through drug-screening, select specific patients for clinical trials, monitor patients during clinical trials, and ultimately work with pharmaceutical companies on the approval of companion diagnostics for precision medicine.
There is significant potential value to be realized by pharmaceuticals companies in the form of efficient identification of new drugs, targeted clinical trials, objective real-time feedback during clinical trials, and expedited regulatory approval by way of companion diagnostics.
Which is precisely why 3DS is focusing on already approved products and late-stage drug trials to commercialize companion diagnostics.
In fact, as I just mentioned, discussions are already underway with several major pharmaceutical companies.
Don’t be surprised to see 3DS announce a major partnership with a big-name pharma company in the near future.
The potential for what 3DS real-time ex-vivo feedback can do for both patients and Big Pharma is massive.
It’s precisely why the Company has attracted some of the top minds in the industry.
I could write a full report on the list of who’s who involved in 3DS, but here’s a brief example of the credentials from the management, board, and advisors – together, they combine decades of experience in diagnostics, healthcare, pharmaceutical and business.
- Business development and marketing work at some of the world’s largest pharmaceutical companies, including Germany’s Bayer AG and US-based Johnson and Johnson.
- Professor of Physiology and Pathophysiology, Biochemistry and Medical Genetics, Human Anatomy and Cell Science, University of Manitoba. Director of The Genomic Centre for Cancer Research and Diagnosis (GCCRD) at University of Manitoba. Internationally known researcher with more than one hundred publications related to research on Genomics and Genomic Instability. Recipient of several academic awards including the Braidwood Jackson Memorial Award; the Dr. Saul Highman Memorial Award; the Rh Award (Basic Science); the J&J Cognition Challenge (2013).
- Chairman of the Centre for Imaging Technology Commercialization. Former Director General of the NRC Institute for Biological Sciences, Ottawa, ON, and founder and Director General of the Institute for Biodiagnostics, Winnipeg, MB. 2008 Outstanding Achievement Award of the Public Service of Canada. Awarded the Queen’s Gold (2002) and Diamond (2012) Jubilee Medals for contributions.
- Internationally recognized for contributions to the treatment of prostate cancer, notably for pioneering the adoption of Active Surveillance as a standard aspect of patient care. Widely published uro-oncologist and a Professor, Department of Surgery, University of Toronto, past Chief of Urology, Sunnybrook Health Sciences Centre, Toronto, and Chairman, World Uro-Oncology Federation. Awarded the Order of Canada in 2016 for his contribution to prostate cancer treatment.
How about business advisor Jonathan Goodman, Director & CEO of Knight Therapeutics Inc., who just put CDN$1 million into 3DS?
Then there’s advisor Dr. Heiner Dreismann who is Past President and CEO of Roche Molecular Diagnostics.
I could go on but you get the point. If you want to learn more about the 3DS team, I’ve included it at the bottom of this Letter.
The people backing 3DS is why I believe in this Company, its platform, the science behind it, and the upside it could provide for early shareholders.
Near-Term Catalysts
As you can imagine, such a profound new technology and research has not gone unnoticed by industry experts – even if its under the radar of financial institutions.
As hinted by 3DS, a steady stream of positive news flow is expected including talent acquisition, clinical trial progress, and new business developments.
All of this means that we could witness some big announcements as early as next year that could significantly propel the Company to new heights.
I confidently believe that once the 3DS business strategy rolls out, institutions will want a piece of the pie.
Conclusion
Based on Dr. Sabine Mai’s groundbreaking research, 3D Signatures is the first and only company to understand and quantify normal versus disease state telomere arrangement, turning this into usable, predictable, reliable, and highly valuable information that can improve patient outcomes.
Highly promising and unconventional software platform is already attracting attention from multinational healthcare companies relatively early in the product development process.
What 3DS is doing is truly remarkable.
We rarely see this type of talent, bandwidth, and opportunity in a junior with such a small market cap.
That’s because few people really know about 3DS. The management team aren’t promoters – they’re business operators and science professionals with incredibly deep backgrounds and a track record of success.
They’re doctors who other doctors ask for advice.
This may be a small company now with a small retail investment market, but I don’t expect it to be for long.
Which means the window of opportunity is closing for early investors.
3DS is right on the brink of not only numerous milestones, but it could also soon begin to generate revenue.
It’s why I invested in this Company and why I think many smart people will too.
3D Signatures Inc.
Canadian Trading Symbol: TSX-V: DXD
US Trading Symbol: OTCQB: TDSGF
German Trading Symbol: FSE: 3D0
Seek the truth,
Ivan Lo
The Equedia Letter
www.equedia.com
Disclosure: We’re biased towards 3D Signatures Inc. because the Company is an advertiser. We currently own shares purchased in a private placement. You can do the math. Our reputation is built upon the companies we feature. That is why we invest in every company we feature in our Equedia Special Report Editions, including 3D Signatures Inc. It’s your money to invest and we don’t share in your profits or your losses, so please take responsibility for doing your own due diligence. Remember, past performance is not indicative of future performance. Just because many of the companies in our previous Equedia Reports have done well, doesn’t mean they all will. Furthermore, 3D Signatures Inc. and its management have no control over our editorial content and any opinions expressed are those of our own. We’re not obligated to write a report on any of our advertisers and we’re not obligated to talk about them just because they advertise with us.
3DS Board, Management, and Advisors
JOHN SWIFT, LLB, CHAIR, BOARD OF DIRECTORS
Past Principal Secretary in the Prime Minister’s Office and Chief of Staff, Office of the Leader of the Opposition, Government of Canada. Past board member of GenXys Health Care Systems, Inex Pharma, Ultrasonix Medical Corp. and Neuromed Technologies Inc. Past Chairman of Central City Foundation.
JASON FLOWERDAY, CEO & DIRECTOR
Mr. Flowerday has extensive life sciences leadership experience including over a decade of business development and marketing work for two of the world’s largest pharmaceutical companies, Germany’s Bayer AG and US-based Johnson and Johnson. Other notable positions include executive leadership and entrepreneurial roles with Knight Therapeutics and Pro Bono Bio Inc. Mr. Flowerday was also co-founder and co-owner of both Orphan Canada and RxMedia Healthcare Communications. He is an independent Director of Aequus Pharmaceutical
SABINE MAI, PHD, DIRECTOR AND CHAIR, CLINICAL AND SCIENTIFIC ADVISORY BOARD
Dr. Sabine Mai is currently Professor of Physiology and Pathophysiology, Biochemistry and Medical Genetics, Human Anatomy and Cell Science, University of Manitoba. She is also Director of The Genomic Centre for Cancer Research and Diagnosis (GCCRD) at University of Manitoba. She is an internationally known researcher who has more than one hundred publications related to research on Genomics and Genomic Instability. Most recently she has contributed to a library of patents related to her work on 3D Genomic Analysis. She is the recipient of several academic awards including the Braidwood Jackson Memorial Award; the Dr. Saul Highman Memorial Award; the Rh Award (Basic Science); the J&J Cognition Challenge (2013). She was recognized in 2015 as one of the Top 100: Canada’s Most Powerful Women.
FERENC SOMOGYVARI, DIRECTOR
Co-founder and former GM of Carl Zeiss Microscopy with more than 38 years in the optical medical and biology industries. Former CEO of 3D Signatures.
IAN SMITH, PHD, DIRECTOR
Dr. Smith, OC, PhD, DSc, FRSC, is currently the Chairman of the Centre for Imaging Technology Commercialization. His past research and commercialization achievements include significant success in the field of magnetic resonance imaging. Dr. Smith is a former Director General of the NRC Institute for Biological Sciences, Ottawa, ON, and founder and Director General of the Institute for Biodiagnostics, Winnipeg, MB. He is a passionate advocate for the advancement of diagnostics for the early detection and treatment of disease.
He was appointed Officer of the Order of Canada in 2008 for his leadership in the advancement, development and commercialization of Canada’s diagnostic technologies, notably magnetic resonance imaging, in the field of health care. In addition, Dr. Smith received the 2008 Outstanding Achievement Award of the Public Service of Canada, presented to individuals who have displayed long-term excellence throughout their careers in Canada’s public service. He was awarded the Queen’s Gold (2002) and Diamond (2012) Jubilee Medals for his contributions.
BRUCE COLWILL, DIRECTOR
Bruce Colwill is a strategic finance professional with over 25 years of experience in start-up and entrepreneurial companies. As the CFO of multiple public and private companies, Bruce has been responsible for fundraising in excess of US$400 million including private and public financing, debt and other structured financings.
GORDON MCCAULEY, DIRECTOR
President and CEO of Viable Healthworks Corp. and Chairman of Life Sciences BC. Co-founder and former President and COO of Neuro Discovery Inc. (NDI Capital). Past President and CEO of Allon Therapeutics.
KEITH B. CASSIDY, CFO
Keith Cassidy is an accountant with significant experience in professional services management (legal), as well as health care and education, at strategic and leadership levels. Keith Cassidy has served as VP Finance and CFO for the Royal Victoria Hospital.
He has held Executive Director positions with several major law firms including Davies Ward Phillips & Vineberg LLP; McMillan LLP; and Bennett Jones LLP. He has also lectured in Business Administration at John Abbott College in Montreal.
OMAR SAMASSEKOU, MD, PHD, VP, CLINICAL PROGRAMS
Oumar Samassekou, PhD, is trained in medical genetics, cytogenetics and other molecular genetics. During his Ph.D. and post-doctoral trainings, Dr. Samassekou developed expertise on prenatal diagnosis and cancer genomics. He has worked on the development of a non-invasive diagnostic procedure to detect fetal chromosomal abnormalities from maternal peripheral blood.
In regards to his background in cancer genomics research, Dr. Samassekou has studied the length of individual telomeres, telemetric nuclear architecture, and genomic instability in leukemia and breast cancer. In addition to his competencies in research, he has been trained in a clinical laboratory examining both molecular genetics and cytogenetics. Dr. Samassekou was the supervisor of a molecular pathology unit in one of the largest laboratories specializing in clinical pathology of breast cancer in Canada. Dr. Samassekou has also developed competency to supervise analytical and clinical validation of different clinical diagnostic, prognostic and predictive tests.
HUGH ROGERS, LLB, VP FINANCE
Mr. Rogers is an entrepreneur and lawyer with broad private and public company experience in business management, regulatory compliance, finance and investor relations. Recent work includes corporate finance advisory positions in a range of industries from health sciences and agribusiness to mining and oil and gas. Recent experience also includes corporate restructuring under the Companies’ Creditors Arrangement Act and disposition of distressed assets. Mr. Rogers holds a B.Sc. and LLB. He is a member in good standing of the Law Society of British Columbia.
CLINICAL AND SCIENTIFIC ADVISORY BOARD
Laurence Klotz – MD, FRCS(C)
Dr. Klotz is internationally recognized for his contributions to the treatment of prostate cancer, notably for pioneering the adoption of Active Surveillance (or “Watchful Waiting”) as a standard aspect of patient care. Dr. Klotz obtained his medical degree and residency training from the University of Toronto with a special fellowship in uro-oncology and tumour biology at Memorial Sloan Kettering Cancer Centre, New York. He is a widely published uro-oncologist who serves on the board or heads many medical/scientific organizations. He is a Professor, Department of Surgery, University of Toronto, past Chief of Urology, Sunnybrook Health Sciences Centre, Toronto, and Chairman, World Uro-Oncology Federation. Dr. Klotz was awarded the Order of Canada in 2016 for his contribution to prostate cancer treatment.
Hans Knecht – MD, FRCPC, FMH, FAMH
Dr. Knecht established himself as a prominent haematologist through his ground-breaking translational research on lymphoma biology. His current focus is on the molecular events leading to the transition from the mononuclear Hodgkin to the multinuclear Reed-Sternberg cell and the impact of 3D nuclear telomere organization on this transformation. Dr. Knecht received his medical degree from the University of Zurich, Switzerland with post-graduate work under both Maxime Seligmann (Haematology) and Karl Lennert (Haematopathology) in Paris and Lausanne, respectively. Dr. Knecht is currently a Professor of Medicine and Chief, Division of Haematology at McGill University and Jewish General Hospital, Montreal.
Darrel Drachenberg – BSc, MD, FRCS(C)
Dr. Drachenberg is a urologic oncologist and researcher and strong proponent of Active Surveillance for prostate cancer patients. Dr. Drachenberg attended medical school at the University of British Columbia and urology residency at Dalhousie University. He is an American Foundation of Urology Scholar with fellowship training in urologic oncology at the National Cancer Institute in Bethesda, Maryland. He founded the laparoscopic urology program and prostate brachytherapy, cryotherapy, and HIFU programs at the University of Manitoba where he works as assistant professor of surgery and director of research for the Manitoba Prostate Center and Section of Urology and Chair of the Genito-Urinary disease site group, CancerCare Manitoba.
Dr. Thomas Cremer – Professor emeritus of Anthropology and Human Genetics, Ludwig Maximilians University, Munich, Germany, Independent Expert
Dr. Cremer is an internationally-recognized scientist specializing in the studies of nuclear architecture. He is one of the pioneers of interphase cytogenetics and comparative genomic hybridization (CGH). These methods have become widely used tools for cytogenetic analyses of chromosomal imbalances.
He is a corresponding member of the Heidelberg Academy for Sciences and Humanities since 2000, a member of the Leopoldina since 2006, a honorary member of both the European Cytogenetics Association (ECA) and the German Society of Human Genetics since 2011, as well as the recipient of the medal of Honor of this Society.
Kenneth C. Anderson, MD
Program Director, Jerome Lipper Multiple Myeloma Center and LeBow Institute for Myeloma Therapeutics Institute Physician, Kraft Family Professor of Medicine, Harvard Medical School
Dr. Anderson graduated from Johns Hopkins Medical School, trained in internal medicine at Johns Hopkins Hospital, and completed hematology, medical oncology, and tumor immunology training at Dana-Farber Cancer Institute.
He serves as chief of the Division of Hematologic Neoplasia, director of the Jerome Lipper Multiple Myeloma Center, and vice chair of the Joint Program in Transfusion Medicine at Dana-Farber.
BUSINESS ADVISORS
Jonathan Goodman, Director & CEO, Knight Therapeutics Inc.
Prior to Knight, Mr. Goodman was the co-founder, President and CEO of Paladin Labs Inc. which was acquired by Endo for $3.2 billion. Under his leadership, $1.50 invested in Paladin at its founding was worth $142 nineteen years later. Prior to co-founding Paladin in 1995, Mr. Goodman was a consultant with Bain & Company and also worked in brand management for Procter & Gamble. Mr. Goodman holds a B.A. with Great Distinction from McGill University and the London School of Economics with 1st Class Honours. Additionally, Mr. Goodman holds an LL.B. and an M.B.A. from McGill University.
Dr. Heiner Dreismann, Past President and CEO, Roche Molecular Diagnostics
Dr. Dreismann is a seasoned executive with more than 24 years experience in the healthcare industry, and is regarded as a pioneer in the early adoption of the polymerase chain reaction (PCR) technique, one of the most ubiquitous technologies in molecular biology and genetics research today. He had a successful career at the Roche Group from 1985 to 2006 where he held several senior positions, including President and CEO, Roche Molecular Systems, Head of Global Business Development, Roche Diagnostics and Member of Roche’s Global Diagnostic Executive Committee. Dr. Dreismann currently serves on the boards of several public and private health care companies. He earned a master of science degree in biology and his doctor of philosophy degree in microbiology/molecular biology (summa cum laude) from Westfaelische Wilhelms University (The University of Munster) in Germany.
John Lindsay, Founder, SciPartners
Mr. Lindsay began his career at Millipore Corporation, Merck KGaA, and quickly advanced to become the youngest Vice President in the history of the company. He was promoted to Executive Vice President of several divisions, including the Analytical Group and Milligen Biosearch Divisions. In 2000, he founded SciPartners, with the objective of building a platform for development of early stage European and North American firms. His focus is the Life Science market, and over the past 14 years he has successfully built up sales and marketing that led to rapid growth and increased revenues for many companies, and the acquisition of ProXeon by ThermoFisher and the acquisition of Halo Genomics by Agilent.










I don’t believe you!show readers and me your deposited cancelled check used to purchase 3D shares.
Very interesting, I took the time to read half the article this morning and the other half now… Stock went up 29% today…
My son was diagnosed with leukemia (ALL) in July 2016, he is 20yrs young. After the initial 4 wk in patient he was released for out patient treatment and put on an aggressive regiment, one that is not offered to many cancer patients because it will kill them. After his 5th week of chemo, he was told that there was no sign of cancer in his blood or system, yet they want him to stay on this 6 month treatment regiment. He has done very well through all this, not your average cancer person at all. But now in his 3 & 4th phase of treatment he is very ill. Twice now his body is free of cancer, we are told. I have wondered from the start, just how many people are free of cancer after 6 weeks but 4 months down the road die? It is not the cancer that kills them, it is the treatment. Something is just not right, like how much do the DR’s get paid and are reimbursed to purchase and then use the chemo meds by the pharmacies? For incentives? Check that out. A DR. That can’t afford the meds will be more likely to talk about this then one that can afford it. It is all about the fucking money. Not about saving a life. One weekend at hospital the on call Dr. Says to us, “you know cancer is tuff, but a 20 yr old life is well worth saving, one that is 62, well not so much.” Wow, wtf? Really!
Venus-My dear son passed away in September after 11 months of chemo… His doctors said his Leukemia was in remission months ago but his body was was failing from the treatment. Kevin was only 45 years old & led a healthy lifestyle, was never I’ll his entire life & is greatly missed. I highly recommend seeking alternative treatments so your son will not have the same outcome…
Would someone discuss death caused by over-treatment of radiation (lung cancer).
Chronic Cerebrospinal Venous Insufficiency CCSVI/Neurovascular Disease
CCSVI is a Treatable Congenital Scientifically Confirmed Recognized Medical Condition, Science Established Causative Factor in Multiple Sclerosis, AND plays a part 43 other so called Neurological afflictions!
CCSVI may be effecting yourself right now and you are unaware!
I am deeply invested in this great company but I wonder for how long this investment is “dead money”. the range between 0.75 and 0.90 starts to get boring.
Ivan could you please explain the formula used to derive at 96 a share. Trying to work out numbers but not sure what you did.
There is absolutely no interest in this company. Very low volumes translates in a absolutely lack of interest in this company. Today we are already 10% lower that the last PP.
DXD needs some strong promotion. Not impressed with the IR people.
Why doesn’t ivan respond to any comments? What is the point of having an option to comment?
I apologize for not responding sooner.
I try my best to keep in contact and respond to readers, but it is very difficult to respond to every inquiry we receive as we have hundreds of thousands of readers per month.
I hope you understand and to not take that as my lack of dedication to readers of this Letter.
Again, my apologies.
Yes, there appears to be some that are letting go of their shares (not sure where this selling pressure is coming from, since most of the $0.35 private placement has been free trade for some time).
However, my view remains the same: I am bullish and not only added to my position since this letter came out, but now own the majority of my position above the current share price.
The Company is making progress, but healthcare stocks take time.
I believe the near-term upside shoot here is if DXD announces a major partnership with a BIG Pharma Company. Unfortunately, this isn’t something we can time so we must be patient if we believe management can accomplish such a feat.
The telomere science surrounding the analysis of certain diseases is truly ground-breaking. It will take time, but could be very rewarding.
I am patient here, but I understand that every one has a different philosophy.
I hope this helps.
Ivan
I phoned couple weeks ago the vice-president of one of the major investment banks in Brazil, the guy was nice and open and I ”pass the ball” to people responsible to 3D, … they said (3D) wasn’t worth to call them (maybe a 20 dollar call to get in return thousands or even more) … I realized that I was working for nothing. C’mon is not everyday I talk with vice-presidents in order to promote a Canadian company. Yet those guys are always busy and don’t bother to answer calls from strangers. I could have done better. I believe totally in 3D and I have invested some bucks on it (too bad I don’t have 50k or more to do it) … surely they will go far, brilliant minds behind it. Cheers Equedia! You guys are awesome.
Thank you for the reply. Didn’t realize the extent of reader traffic and the demand that puts on you however the professional onus is on you and your professional peers to make sure your readers are treated with respect and in timely manner. I noticed a few comments that were directed towards you specifically and so I felt compelled to get you to respond.
I do agree with your perspective on patience here, and some sort of partnership which will serve as a catalyst for the share price take off.
Regards and I hope you have an awesome time on April 4th!
745317 463695 You really should take part in a contest for among the very best blogs on the internet. I will recommend this internet site! 589626
I also believe strongly in DXD and it’s technology but I am quite disappointed in the lack of interest in this company. I think the IR people have to work a bit harder. i am getting tired to see prices in the 0.60’s.
Good luck to all and I thank Equedia for their recommendations
Since you disclosed your cheque to them wouldn’t it be transparent to disclose their cheque to you?
Just saying…since you make it appear that 50k is a significant commitment. But once your reader knows that it’s much less then what they paid you to promote it the significance of your investment slowly diminishes.
Regards,
Ivan, I’m waiting for your disclosure on how much you received from them. Thank you, (if not I will unsubscribe)
I do agree with SRDAN. But what I like about Equedia is that he always answers my questions,
But the stock market, specially the TSX venture, is a big circus with a lot of clowns involved.
So for me Equedia is the good clown, but a clown it is
Would love some feedback from Ivan regarding the above as well as thoughts now and rumors of terrible management at the company – or if it’s simply a 50% off better deal than from the time this article was written.
Nevermind – here we are: “Equedia Network Corporation., owner of Equedia.com has thus far been paid $160,000 plus GST of advertising coverage for 3D Signatures on equedia.com plus any additional expenses we may incur as a result of additional advertising. 3D Signatures has paid for this service. This contract has been extended by an additional $150,000. We currently own shares of 3D Signatures purchased in two separate private placements and we may purchase shares of 3D Signatures without notice and intend to sell every share we purchase for our own profit.”
Thanks Adrian. Ivan is shady at best, captivating writer nonetheless! I no longer receive his letters.
Regards
Thanks for the comments.
Guys, we promote companies we like and help them gain exposure because without exposure, public small cap companies just don’t stand a chance.
I wouldn’t invest in any small cap company that doesn’t have an IR marketing budget and I hope our track record speaks for itself.
As for our interest, we still own a large position in 3D and are hopeful the Company will turn things around. Great tech, just needs to be better managed.
Hope this helps.
Ivan
Dear Ivan,
It’s good to know that you still own a large position in DXD. It’s a shame that management didn’t change. That said, I think there are some great NR’s in the pipeline once the PP is closed, hopefully at November 15.
But I don’t think they will have a budget for marketing. Like you said that is very troublesome. Are you still going to promote DXD after good news even when you don’t receive money from them? Ask for shares. With this technology, it can be a great investment
Thank for all the info and professional website.
Luc Janssens
Intent to file for bankruptcy on May 31…..Not a great recommend! There goes your $50K